Nature of Biofuels
Biofuel: A Net-negative Pathway
- Biofuels are way to take out CO2 from the landmass above the surface of the earth and putting it back in atmosphere.
- This is unlike fossils where you are taking carbon stock from below the surface and putting it in atmosphere increasing its concentration.
- Besides the amount of CO2 you are putting by burning 1 kg of biomass is much lesser than the amount of CO2 the plants used to make 1 kg of plant biomass. (plants convert only 0.5% of photosynthesis to biomass)
- Thus, biofuels are net-negative in terms of carbon emissions.
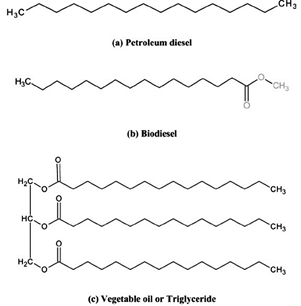
As Hydrocarbon: Biofuel v/s Fossil
- Biofuel is small ring hydrocarbons with lots of oxygen and benzene rings. (not suitable remember)
- Fossil as we have seen is just biomass buried for millions of years under low oxygen conditions.
- Besides biomass is long polymers of sugar molecules.
- So, if we somehow remove oxygen and break the polymer, we can get biofuels.
Biomass as Energy Store
- Further biomass is basically solar energy trapped in the form of chemicals that make up the living beings.
Biomass Composition
- C 50%
- O 40%
- H 5 %
- N 1%(From protein)
- P 0.5% (From protein)
Biomass Constituents
- Primarily made of sugar polymers with lots O2 and closed ring hydrocarbons. (half the plant biomass is fundamentally sugar)
- Plant cells are made up of cell walls that gives a protective layer to help plants stand tall.
- The cell wall is made of microfibrils of cellulose which is basically sugar polymer. (1/2 of cell wall)
- At later stages there is an additional protective layer in the form of hard cover in trees which is called lignin.
- Lignin is plastic-like. It is not degradable, has lots of aromatics.
Classification of Biofuels

- For the sake of simplicity think of plant biomass in terms of edible and non-edible part.
- The edible part of plant like fruits, vegetables, grains etc. are usually made simple sugar, starch.
- The other edible part is seeds that have edible oils.
- The non-edible part of biomass on the other hand is made of cellulose, hemi-cellulose and lignin.
Energy From Biofuels
- There are 3 pathways to derive energy out of biofuels:
- Burn them
- Make alcohols like ethanol and methanol
- Make biodiesel
- Make biogas
Burning of Biofuels
- Burning solid biofuel will leave solid ash residue which is not desirable. Eg: Cow dung, wood etc
- So, we need to gasify or liquify biomass.
Liquifying Biofuels
- Liquifying biofuels again include 2 pathways
- Make alcohols: ethanol and methanol: fermentation
- Make biodiesel: From vegetable oils: transesterification
Gasifying Biofuels
- Biogas: Bio-methanation
- Biohydrogen: from syngas
Alcohols: Ethanol and Methanol
- Alcohols are best alternatives to petrol.
- You can directly replace petrol with ethanol or methanol in IC engines or alternately you may blend it with petrol. (Ethanol blending upto 20% by 2030 is the target)
How are they made?
Fermentation
- Alcohols are made by fermentation of sugars by microorganisms.
- Depending on the microorganism used and sugar source you get different alcohols.
Ethanol
- Most commonly formed alcohol through fermentation is ethanol.
- Fermentation is simply breaking down of sugar molecules in the absence of oxygen at the cellular level or in other words anerobic cellular respiration to produce ethanol. (For detailed discission on fermentation see the optional section below)

Which microorganisms produce ethanol?
- Microorganisms capable of producing ethanol include yeast, some species of bacteria, fungi and some species of micro algae.
Feedstock
Edible part
- Ethanol can be made from both edible parts and non-edible cellulosic biomass.
- Edible sources include sugarcane, corn, and other starch-based crops (sugar beets, rice, wheat, potatoes)
Non-edible part
- The cellulosic matter from plant biomass are basically complex sugar molecules.
- Thus, they have to be broken down into simple sugar in order to be fermented into alcohols.
- In order to break them down hydrolysis is done in other words treatment with water. These simple sugars can then be used for fermentation by microbes to produce ethanol.
- However, lignin is made of phenols and not sugars. Thus, they cannot be converted into alcohols.
Advantage
- No SOX emissions
- Can be used as a substitute for petrol in SI engines
- Low carbon emissions (biofuels are net negative)
- Low NOx emissions
Methanol
- Although methanol can also be made through fermentation, it is rare.
- This is because it is produced in small amounts as by-products during fermentation of certain bacteria and fungi species.
- Thus, the commonly used pathway to make methanol is producing syngas. (discussed in alternate fuels: Gas-to-liquid section)
Optional section
Photosynthesis and Cellular respiration: the universal energy pathway

Photosynthesis: building the energy stores
- Plants have simply collected energy from sunlight and stored it in sugar molecules through the process of photosynthesis.
- Under photosynthesis, photoreceptors in plants called chlorophyll absorb sunlight. When light falls on chlorophyll it excites electrons in the chlorophyll. This is how energy from sunlight is captured.
- The excited electron then carries this energy and transports it to different parts of the plant body and stores. (follow the path of electron in the figure)
- The energy carried by the electrons is stored in a kind of rechargeable batteries in plants called ATP molecules. (future civil servants need not know the details of this molecule)
- These rechargeable batteries called ATP are stored in the sugar molecules of plant biomass built by plants.
- To do this, plants use CO2 and H2O in the atmosphere.
Cellular respiration: using the energy stores
- Ultimately all living organisms access energy through the process of cellular respiration.
- The process of utilizing the energy contained in food is called cellular respiration.
- Cellular respiration is just a way of taking in energy stored in the sugar molecules to run life activities.
- The food we eat is first digested or broken into simple sugars and carried in the bloodstream which carries the energy-rich sugar to all the cells of the body.
- The cells in turn use the energy in the sugar to form the rechargeable batteries ATP in the cells which then acts as drivers of life activity. Once done CO2 is released.
- Again, it is the electrons that carry energy.
- Once all the energy is delivered to ATP the electron needs a place to go. For this all living beings have electron acceptors.
- Oxygen is the most common electron acceptor. That’s why we breathe oxygen.
- Finally, after oxygen receives electrons, it combines with hydrogen to form water completing the process of cellular respiration.
- Thus, for cellular respiration we need glucose and oxygen.
- C6H12O6 + O2 à CO2 + H2O + energy (needed for life activities)
Cellular respiration in low oxygen condition
- When we are doing strenuous job like running or swimming, we need more energy. This energy is got from breaking down of glycogen (storehouse of glucose in our body) that is stored in the liver.
- However, there is not enough oxygen to accept electrons. Thus, cellular respiration comes to a halt.
- Alternately humans use pyruvate molecules (broken sugar) as electron acceptor instead of oxygen.
- When pyruvate accepts electron, it forms lactic acid instead of H2O.
Fermentation: cellular respiration in the absence of oxygen
- Yeasts like humans also breakdown their food using oxygen.
- In the absence of oxygen, they produce alcohols that we drink.
- In yeasts after breaking down of glucose into pyruvate. After this the pyruvate is converted into acetaldehyde which acts as electron acceptor in the absence of oxygen.
- When acetaldehyde accepts electron, it forms ethanol.
- This anerobic respiration is called fermentation.
Biodiesel: From Transesterification
What is Biodiesel?
- Oils from oilseeds are basically straight chain hydrocarbons but long ones.
- This is the only difference between biodiesel and diesel.
- If we take vegetable oil and break its long hydrocarbon straight chain into 1/3rd, 1/3rd, 1/3rd you get biodiesel.
- Biodiesel can directly replace diesel in diesel-IC engines.
- The resultant product, i.e, biodiesel is called ester.
- That’s why the process of breaking long straight chain hydrocarbons into short chains is called transesterification.
- Breaking can be done by following ways:
- Heat it: Pyrolysis
- Apply pressure: Cracking
- Replace double bonds with hydrogen: Hydrogenation

Feedstock
- Any oil seed can be used to extract oil. However, better option is to use non-edible oil seeds.
- There are more than 200 variety of oil seeds that can be used to produce biodiesel.
- Some common examples include rice bran, sal, neem, mahua, karanja, castor, linseed, jatropha, honge, rubber seed etc.
Advantages
| Fuel | Energy density] (in MJ/ kg) |
| Ethanol | 24-25 |
| Peirol | 43-44 |
| Biodiesel | 40-41 |
| Diesel | 45.5 |
- High energy density
- Low energy input
- Nitrogen-fixation
- No Sulphur
- No aromatics
A case for biodiesel
- India uses 5 times more diesel than petrol, so an alternative for diesel is more important than that for petrol.
- De-sulphurisation of diesel is cost intensive.
- Rural development: growing oilseed-based crops for biodiesel will augment farmer’s income.
- Converting degraded land
- Improves soil fertility as most oilseed-crops are leguminous crops which helps in nitrogen fixation.
Biofuels: Generations
- Biofuels are classified into different generations based on the strategy adopted to derive energy from biomass.
- Accordingly, there are 4 generations of biofuels.
1st generation (Mostly edible source)
- Biofuels derived from edible source.
- Includes both starch-based ethanol and biodiesel (made from edible vegetable oil)
- Sugarcane, corn, and other starch-based crops (sugar beets, rice, wheat, potatoes)
- Oilseeds include rape-seed, sunflower seed, soybeans, palm seeds, rice bran etc.
- Non-edible source Jatropha in India
2nd generation (cellulosic biofuel)
- Non-edible feedstocks like grasses, leaves etc
- Ligno-cellulosic: Mostly wood
- Agricultural waste including rice husk, corn stalks etc.
3rd generation (mostly algal biomass-based)
Biofuels from high-photosynthetic efficiency organisms like algae.

- Feed-stock include:
- Macro-algaeMicro-algae
- Aquatic plants (water hyacinth)
- While most plants are very inefficient in photosynthesis (only 0.5% of sunlight is captured), algae have very high photosynthetic efficiency capable of growing very fast capturing the CO2 from atmosphere.
- Algae extracts large amounts of the carbon dioxide to feed their growth and very little is left to emit to the open air.
- The so grown algae can be used as input for producing both ethanol and biodiesel.
Advantages
- Fastest photosynthesis
- Do not need arable land
- CO2 sequestration (CO2 source can be coal-based power plants too, see section on Direct capture under CCUS technologies)
- Lipids (can be used for biodiesel) and protein source (single-cell protein source)
Liquid Tree: Liquid 3.0
- Recently in Serbia scientists have created such a photo-bioreactor using algae to capture CO2 from the atmosphere.
- It contains 600 litres of water and works by using microalgae to bind carbon dioxide and produce pure oxygen through photosynthesis, in addition to producing biofuels.
Note on Methanogens
- Additionally, there are organisms called Methanogens which capture sunlight to produce methane.
4th generation biofuels: Solar fuels: Artificial leaf and Photosheets
- This is application of synthetic biology to make biofuels.
- The approach includes mimic the biological process of photosynthesis to make energy-rich fuels. (Artificial photosynthesis)
- Currently there are two types of technologies performing artificial photosynthesis namely artificial leaf which produces syngas and photosheet technology which produces formic acid.
- Artificial leaf is a silicon-device powered by sunlight which produces synthesis gas (SynGas) by capturing CO2 form atmosphere or flue gas from power plants.
- While artificial leaf is a cleaner way to produce syngas their main limitation is its produces gas and takes an extra effort to store it in the form of liquid.

- Photosheet technology on the other hand produces formic acid which is stored as a liquid fuel.
Gasifying Biofuels: Biomethanation and CBG
- Always remember the best way to biomass for energy production is to somehow extract only methane out of it and leave the rest for plant use.
- To gasify biomass is to copy what happens in a cow’s stomach.
- Unlike humans, cow breaks down cellulosic biomass in its digestive process called anaerobic digestion.
- Cows have in its digestive system a large fermentation chamber filled with billions of microbes like bacteria and protozoa.
- The food that enters the fermentation chamber is broken down by these microbes, in the absence of oxygen, producing methane in its burps and fart. Mimic this process using bio-digesters you get biogas.
- Biogas is a mixture of methane, CO2 and hydrogen sulphide. So you need to separate CH4 before using it.
- Once separated compress methane to store it and transport it. This process is called bio-methanation.
- Feedstock can be agriculture waste which is mostly cellulosic biomass, cattle dung, sugarcane press mud, municipal wet waste etc.
SATAT
- In 2018 Sustainable Alternative Towards Affordable Transportation (SATAT) initiative was introduced to promote CBG as an alternative to CNG.
- Target is to produce 15 million tonnes of gas aiming to reduce CNG use by 40%. (CNG is imported)
- Particularly useful in states like Punjab, Haryana and Uttar Pradesh which are infamous for stubble-burning.
